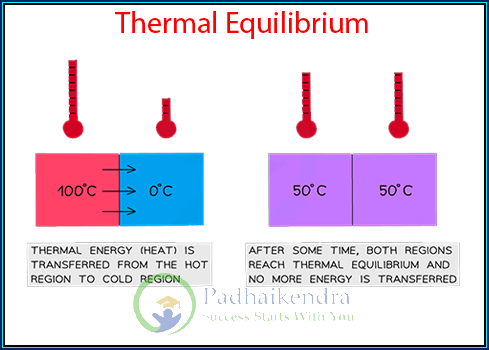
Thermal equilibrium refers to a state in which two or more objects or systems are in thermal contact and have reached a common temperature. When objects are in thermal equilibrium, there is no net transfer of heat between them. This concept is based on the understanding of the second law of thermodynamics, which states that heat spontaneously flows from a higher temperature region to a lower temperature region until thermal equilibrium is reached.
In thermal equilibrium, the objects or systems involved have the same temperature. Temperature is a measure of the average kinetic energy of the particles within a substance. When two objects are brought into thermal contact, their particles interact with each other, leading to the transfer of energy between them. This energy transfer occurs through the process of heat conduction, where higher-energy particles collide with lower-energy particles, transferring some of their energy in the process.
As heat is transferred, the objects’ temperatures may change until they eventually stabilize at the same temperature. At this point, thermal equilibrium is achieved, and there is no further net flow of heat between the objects. The particles within the objects continue to exchange energy through collisions, but the overall average energy and temperature remain constant.
 Thermal equilibrium is an important concept in various areas, including thermodynamics, heat transfer, and the study of thermal systems. It serves as the basis for understanding processes like thermal conduction, convection, and radiation. In practical applications, achieving thermal equilibrium is often desirable for maintaining stable conditions and ensuring accurate measurements and calculations.
Thermal equilibrium is an important concept in various areas, including thermodynamics, heat transfer, and the study of thermal systems. It serves as the basis for understanding processes like thermal conduction, convection, and radiation. In practical applications, achieving thermal equilibrium is often desirable for maintaining stable conditions and ensuring accurate measurements and calculations.
It’s worth noting that achieving thermal equilibrium does not necessarily mean that the objects are in mechanical or chemical equilibrium. Mechanical equilibrium refers to a state where there is no net force or motion between objects, while chemical equilibrium refers to a state where there is no net change in the chemical composition of a system. These concepts are distinct from thermal equilibrium but can be interrelated in certain situations.
Thermal equilibrium occurs when two or more objects or systems in thermal contact have the same temperature and there is no net transfer of heat between them. It is a fundamental concept in thermodynamics and plays a crucial role in understanding heat transfer and thermal processes.
Thermal Equilibrium FAQs
Thermal equilibrium refers to a state in which two or more objects or systems are at the same temperature and there is no net transfer of heat between them. In this state, the thermal energy is evenly distributed, and there is no temperature difference between the objects.
Thermal equilibrium is achieved when two objects or systems come into contact and reach the same temperature through the transfer of heat. Heat will flow from the object with higher temperature to the one with lower temperature until both reach the same temperature.
Thermal equilibrium is significant because it represents a state of balance and stability in terms of thermal energy. It ensures that there is no further heat transfer between objects, and it allows for accurate measurements of temperature and the study of heat flow and thermodynamics.
No, objects in thermal equilibrium must have the same temperature. If two objects have different temperatures, heat will flow between them until they reach the same temperature and establish thermal equilibrium.
For thermal equilibrium to be achieved, there are two main conditions: First, the objects or systems must be in thermal contact, allowing heat transfer between them. Second, there should be no net heat flow, meaning that the rate of heat transfer from one object to another is equal to the rate of heat transfer in the opposite direction.
Thermal equilibrium plays a crucial role in various aspects of daily life. It ensures the effectiveness of temperature measurements using thermometers, enables the proper functioning of heating and cooling systems, and allows for the comfortable regulation of indoor temperature. It is also fundamental in understanding heat exchange in cooking, heating, and cooling processes.
The concept of thermal equilibrium is closely related to the zeroth law of thermodynamics, which states that if two systems are each in thermal equilibrium with a third system, then they are in thermal equilibrium with each other. This law establishes the foundation for the measurement and comparison of temperatures.
In an isolated system, thermal equilibrium can be maintained indefinitely as long as no external heat source or sink is introduced. However, in real-world scenarios, it can be challenging to achieve perfect thermal equilibrium due to factors such as heat transfer through conduction, convection, and radiation.
Thermal equilibrium refers to a state where there is no net transfer of heat between objects, while thermal insulation refers to the prevention or reduction of heat transfer between objects. Thermal insulation is often achieved by using materials with low thermal conductivity to minimize heat transfer and maintain temperature differences.
In an open system, thermal equilibrium can be approached, but it is challenging to achieve completely. This is because open systems allow for the continuous exchange of heat with the surroundings, which can disrupt thermal equilibrium. However, if the system is sufficiently large or well-insulated, it can approximate thermal equilibrium with the surroundings over time.
 Thermal equilibrium is an important concept in various areas, including thermodynamics, heat transfer, and the study of thermal systems. It serves as the basis for understanding processes like thermal conduction, convection, and radiation. In practical applications, achieving thermal equilibrium is often desirable for maintaining stable conditions and ensuring accurate measurements and calculations.
Thermal equilibrium is an important concept in various areas, including thermodynamics, heat transfer, and the study of thermal systems. It serves as the basis for understanding processes like thermal conduction, convection, and radiation. In practical applications, achieving thermal equilibrium is often desirable for maintaining stable conditions and ensuring accurate measurements and calculations.